Chem Unit 6 Homework 1
star
star
star
star
star
Last updated 3 months ago
10 questions
Note from the author:
3
3
2
3
1
3
2
1
1
1
| Draggable item | arrow_right_alt | Corresponding Item |
|---|---|---|
| arrow_right_alt | ||
| arrow_right_alt | ||
| arrow_right_alt | ||
| arrow_right_alt | ||
| arrow_right_alt |
| Draggable item | arrow_right_alt | Corresponding Item |
|---|---|---|
| arrow_right_alt | ||
| arrow_right_alt | ||
| arrow_right_alt | ||
| arrow_right_alt | ||
| arrow_right_alt | ||
| arrow_right_alt |
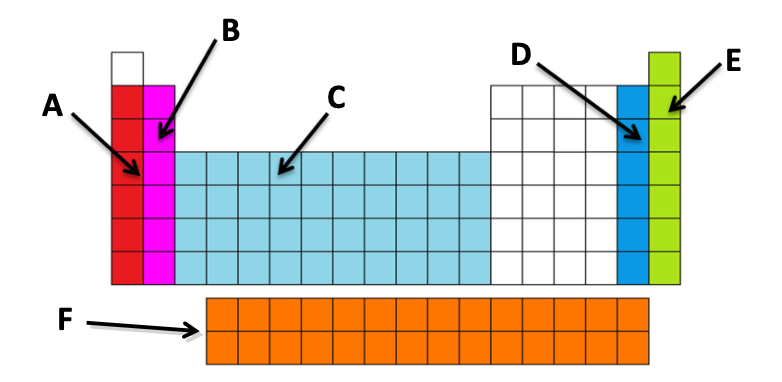
| Draggable item | arrow_right_alt | Corresponding Item |
|---|---|---|
| arrow_right_alt | ||
| arrow_right_alt | ||
| arrow_right_alt | ||
| arrow_right_alt |
| Draggable item | arrow_right_alt | Corresponding Item |
|---|---|---|
| arrow_right_alt | ||
| arrow_right_alt | ||
| arrow_right_alt |