G11 Rates of reactions E-day Project
star
star
star
star
star
Last updated over 1 year ago
37 questions
Note from the author:
Using graphs to determine the rate of a reaction
Required
1
1
1
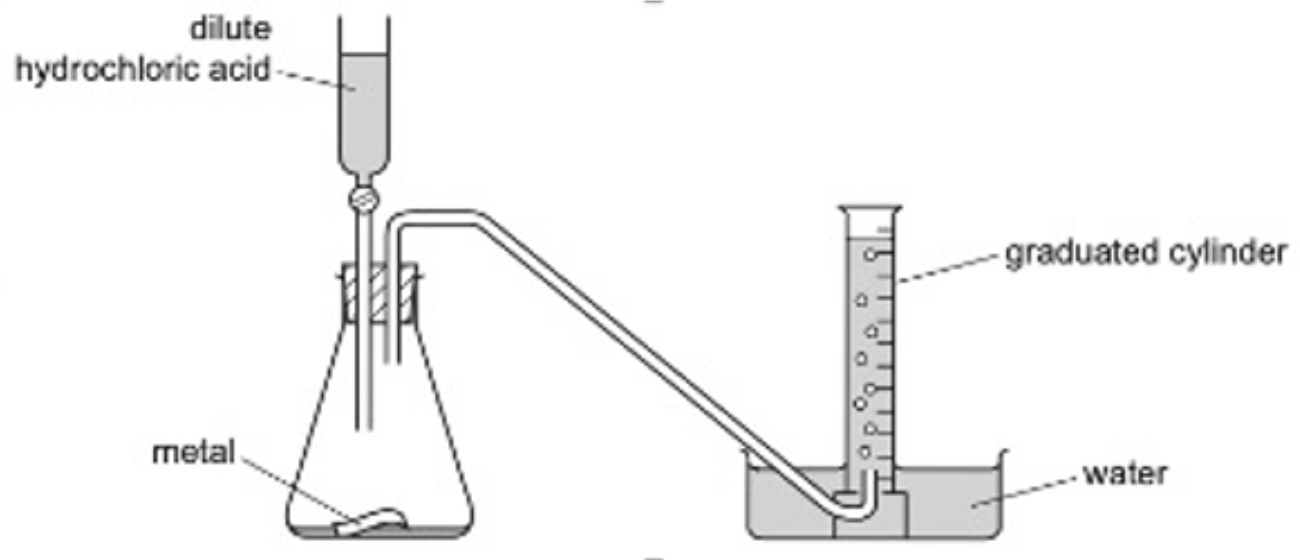
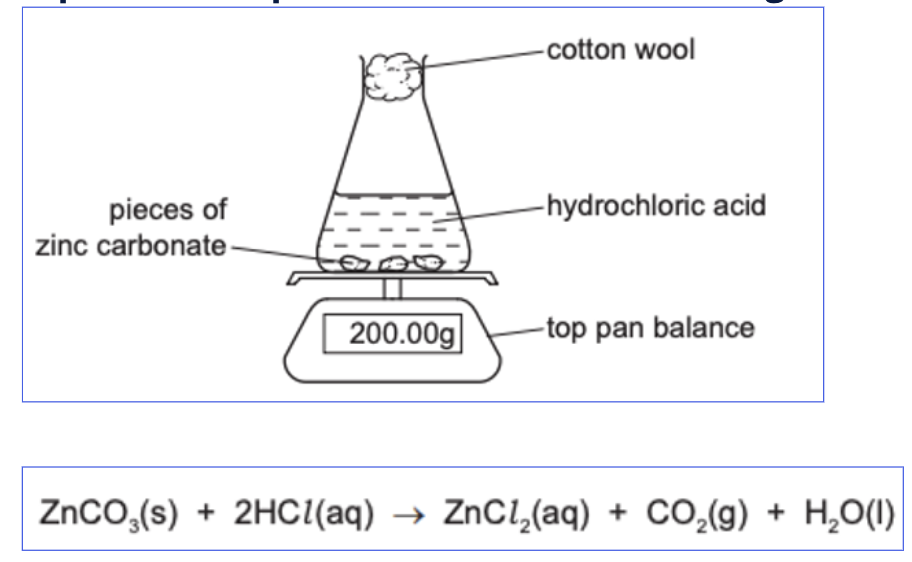
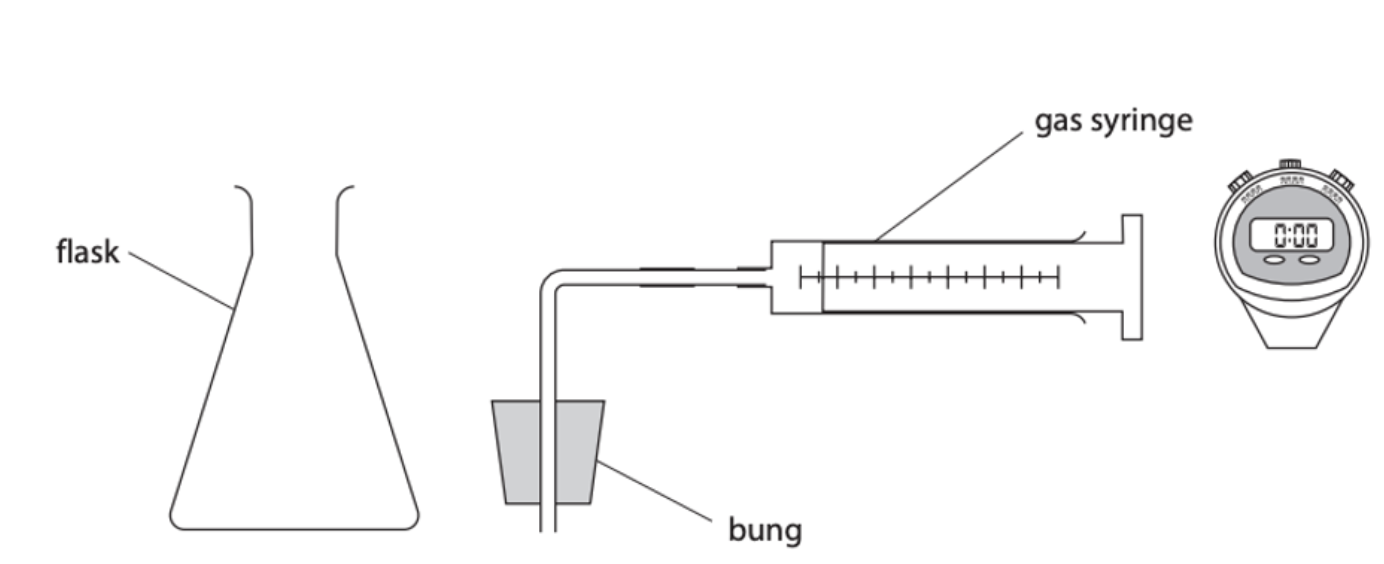
Measuring the rate of reaction in the laboratory
2
1
1
2
Required
1
The factors that affect the rate of a reaction
1
1
1
1
1
1
Required
1
Required
1
General questions on rates of reactions
2
2
1
General laboratory skills
2
2
1