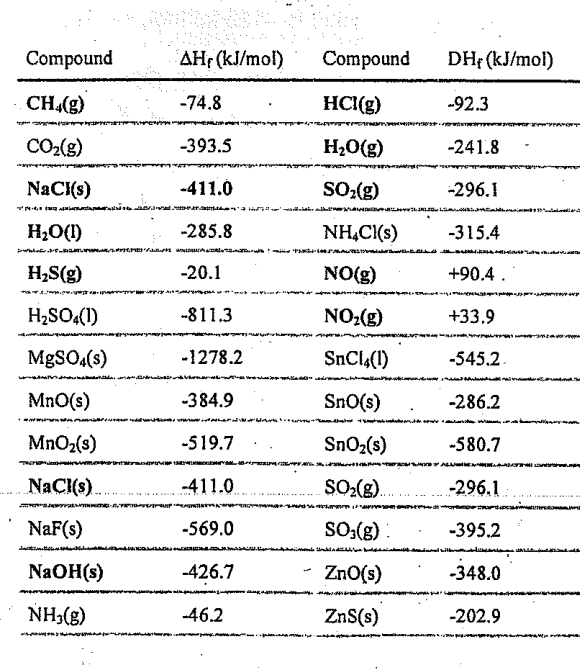
As a class, we will be doing the first two questions together. Complete the rest yourself!
Remember if the standard heat of the reaction is negative, the reaction is exothermic.
Remember if the standard heat of the reaction is positive, the reaction is endothermic.