Quarter 1 Test
star
star
star
star
star
Last updated almost 2 years ago
25 questions
Note from the author:
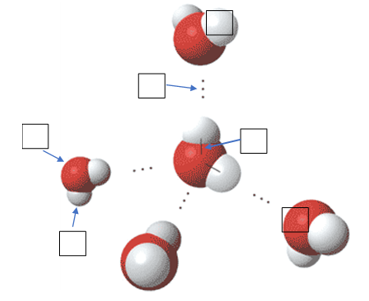
1.1a I can explain how water’s physical properties contribute to polarity, adhesion, cohesion, thermal regulation, density and solubility
1
1.1a
1
1.1a
1
1.1a
1
1.1a
1
1.1a
1.1b I can relate the chemical and physical properties of water to life’s metabolic activities
1
1.1b
1
1.1b
1
1.1b
1
1.1b
1
1.1b
1.2a – I can recognize that living cells are composed of relatively few elements including carbon (C), hydrogen (H), nitrogen (N), oxygen (O), phosphorous (P), and sulfur (S).d Section
1
1.2a
1
1.2a
1
1.2a
1
1.2a
1
1.2a
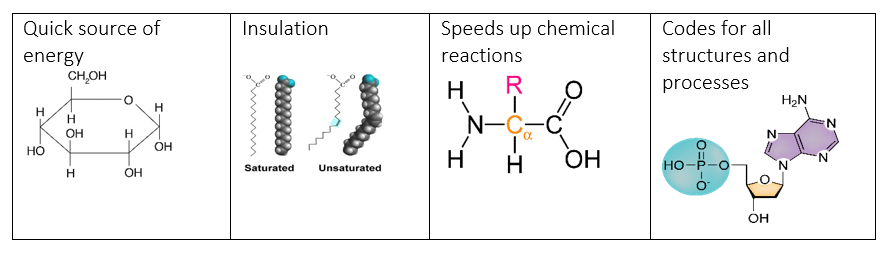
1.2b - I can differentiate between the four major categories of macromolecules (lipids, carbohydrates, proteins, and nucleic acids) through their primary roles and functions.
1
1.2b
1
1.2b
1
1.2b
1
1.2b
1
1.2b
1.3a I can describe the structure of enzymes and explain their in role in acting as catalysts to control the rate of metabolic reactions
1
1.3a
1
1.3a
1
1.3a
1
1.3a
1
1.3a