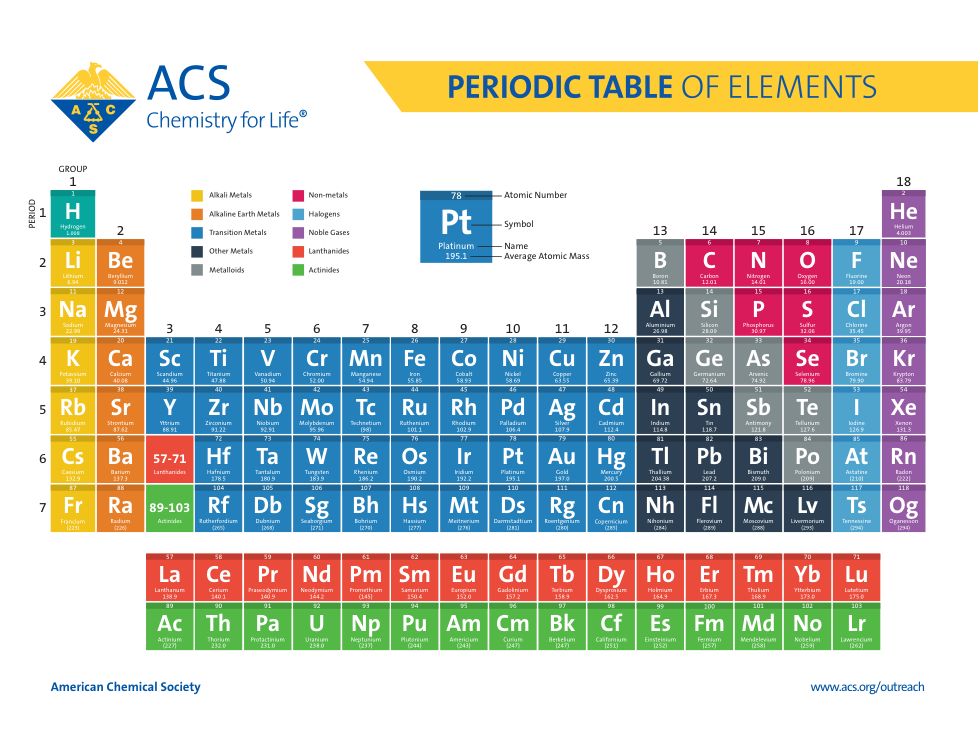
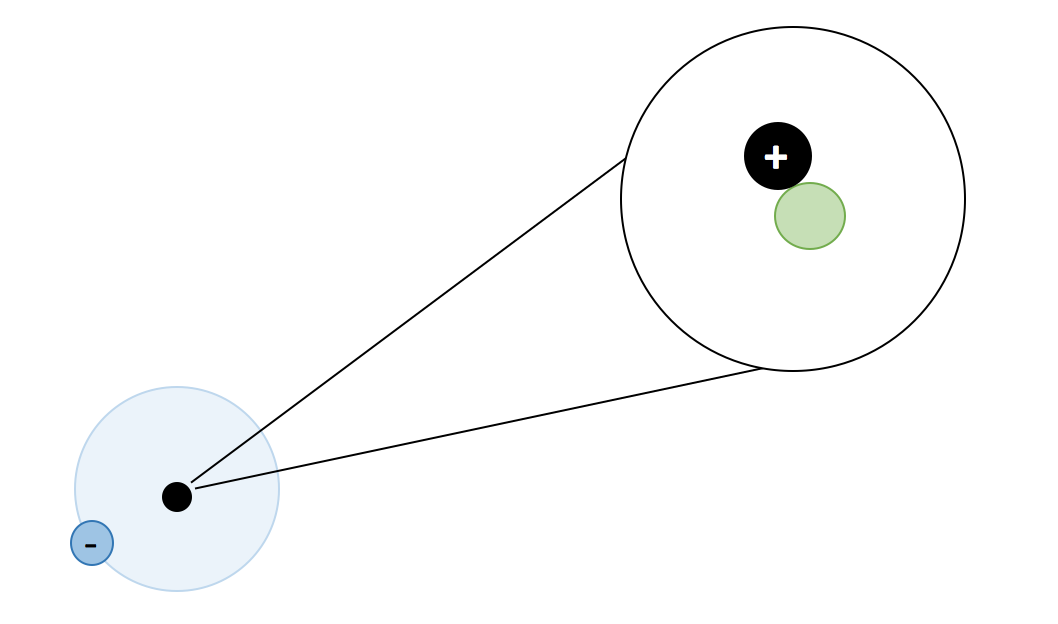
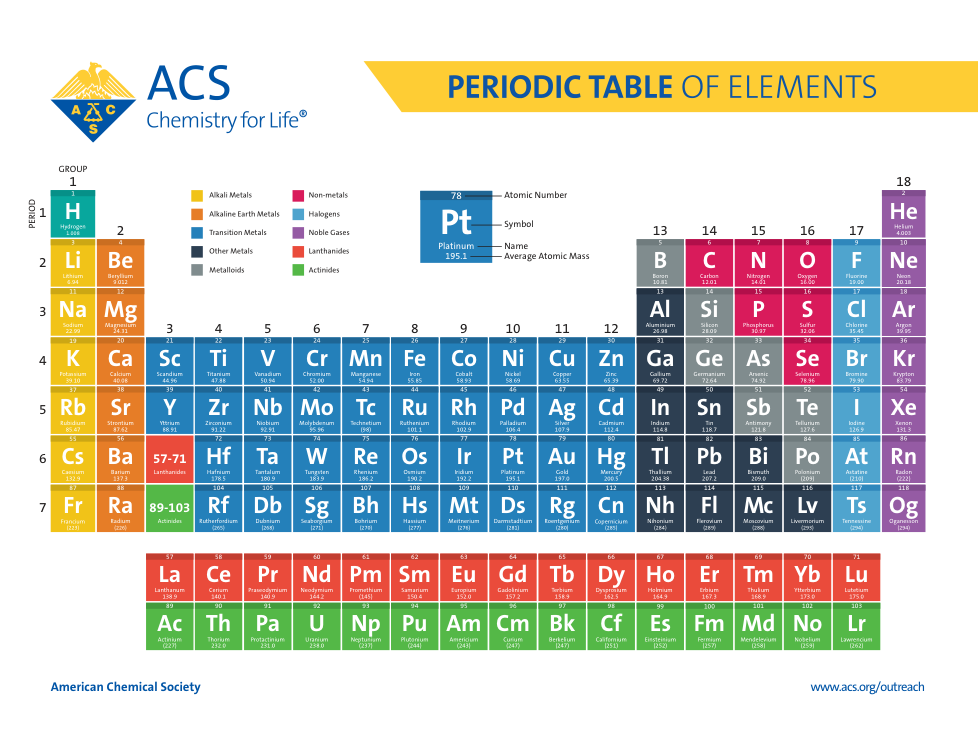
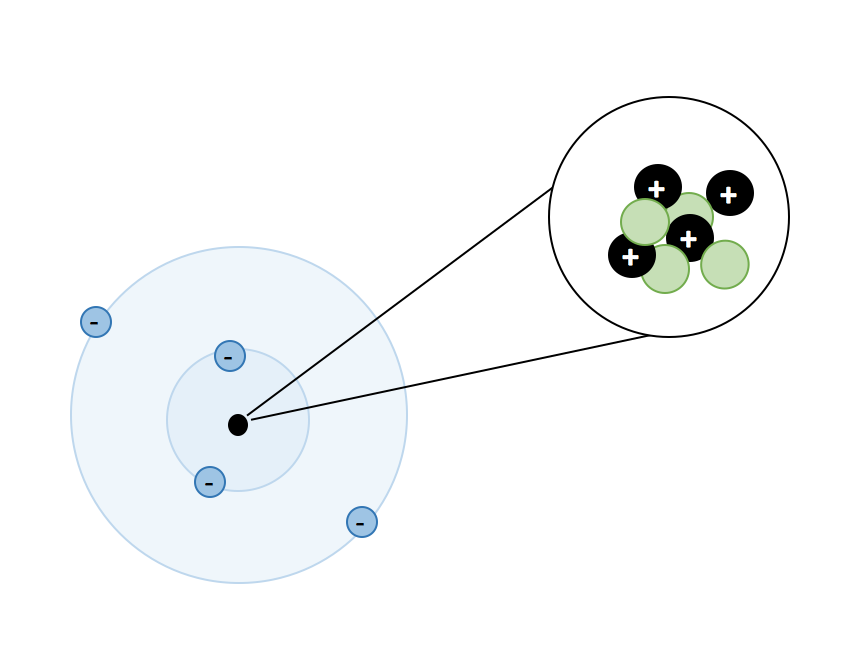
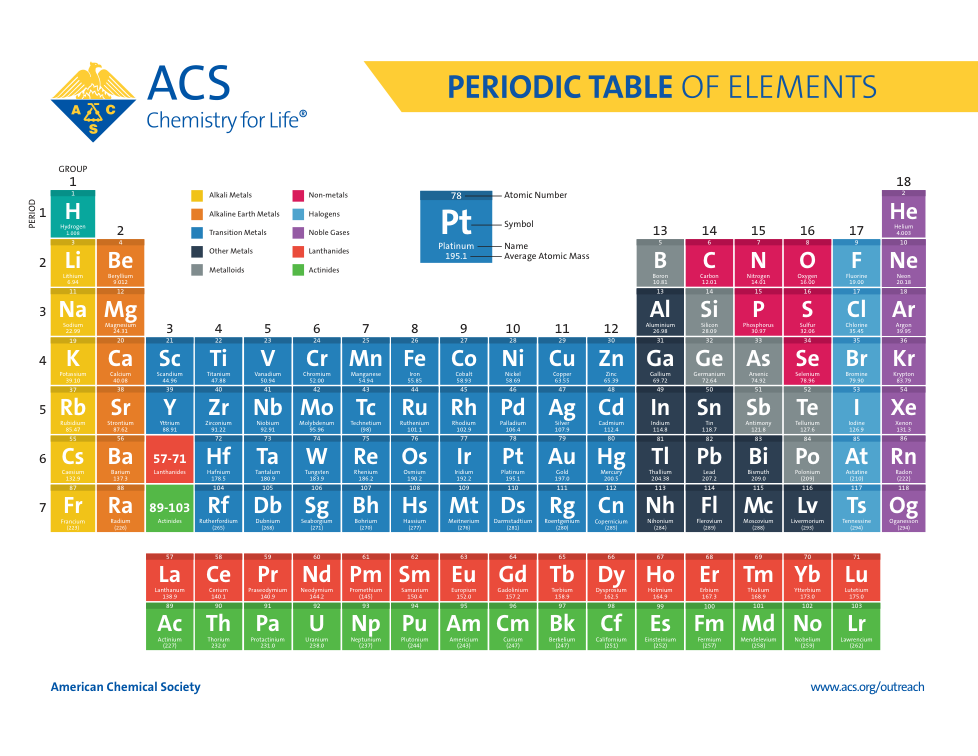
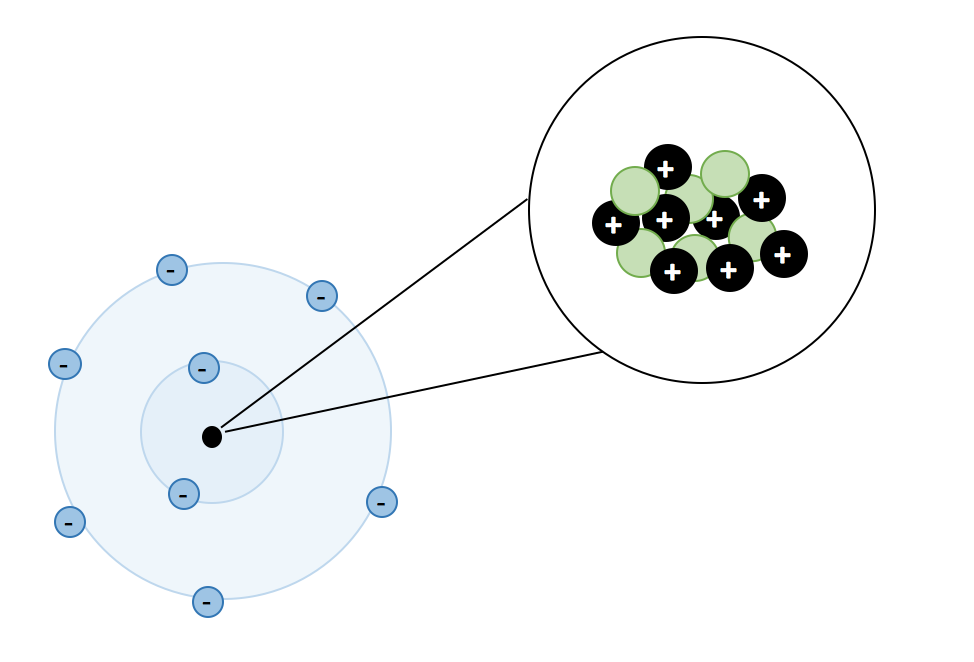
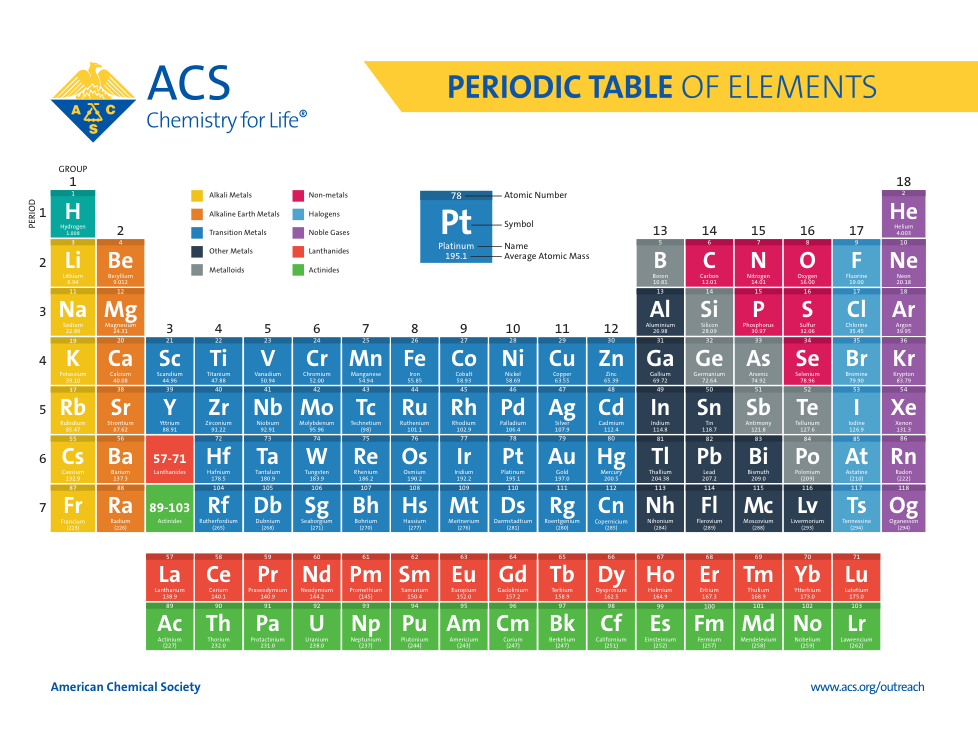
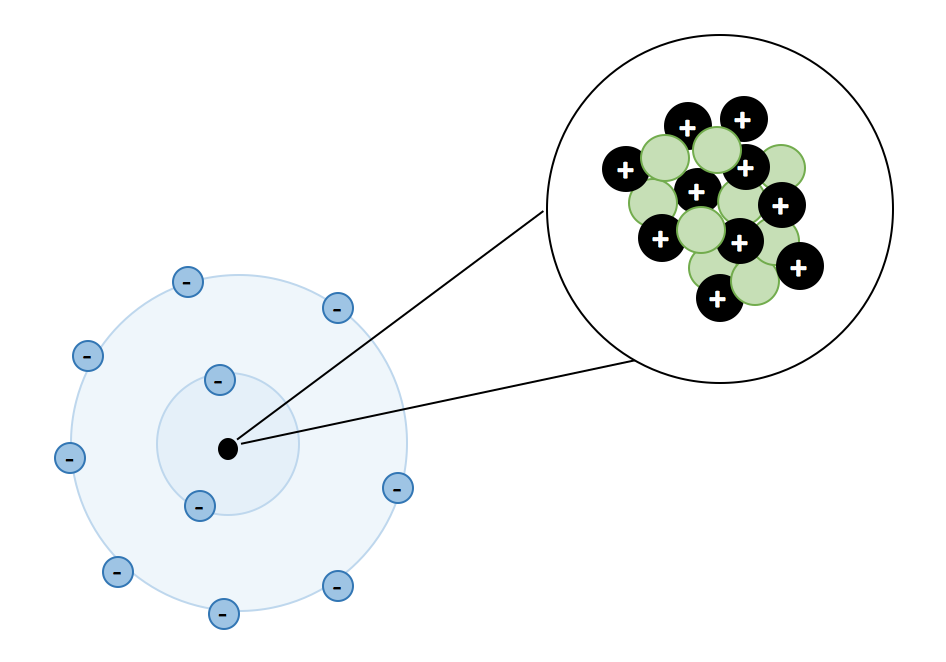
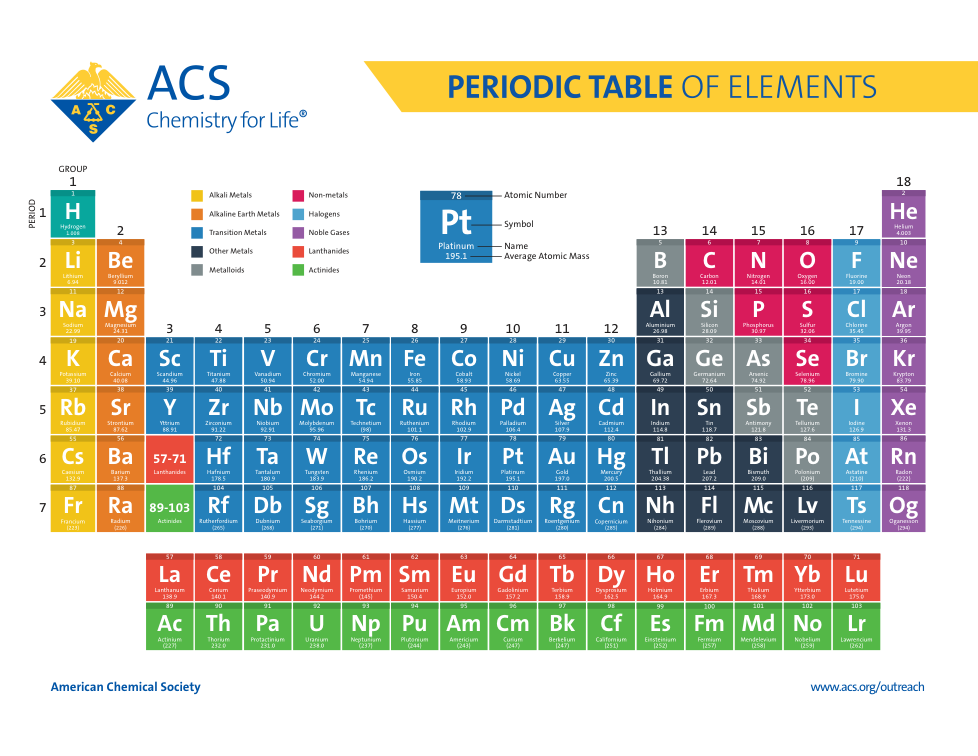
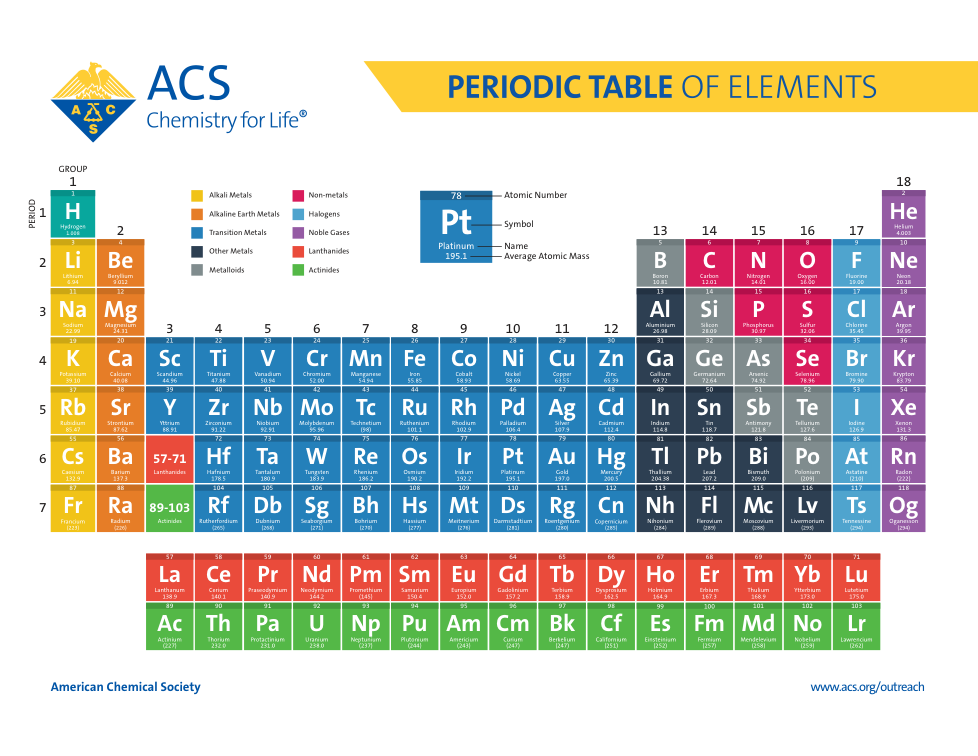
Atoms, Matter, & Elements Quiz (6.P.2.1.)
star
star
star
star
star
Last updated about 2 years ago
20 questions
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1