CP Chemistry Semester 1 Final Exam
star
star
star
star
star
Last updated almost 2 years ago
48 questions
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
5
1
8
3
4
1
3
1
1
6
| Draggable item | arrow_right_alt | Corresponding Item |
|---|---|---|
| arrow_right_alt | ||
| arrow_right_alt | ||
| arrow_right_alt | ||
| arrow_right_alt | ||
| arrow_right_alt |
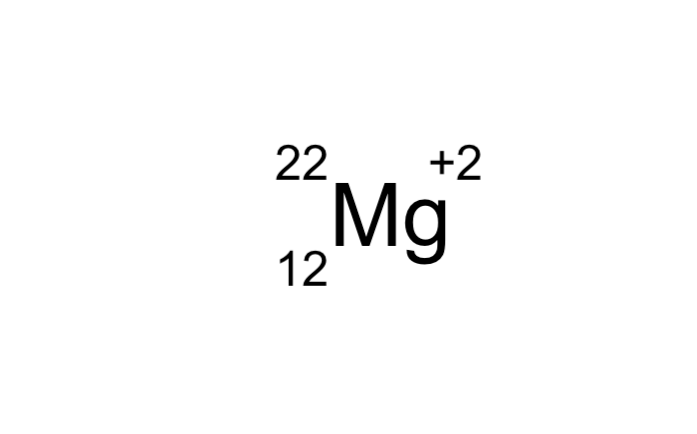
| Draggable item | arrow_right_alt | Corresponding Item |
|---|---|---|
| arrow_right_alt | ||
| arrow_right_alt | ||
| arrow_right_alt |