Atoms and Molecules
star
star
star
star
star
Last updated over 1 year ago
14 questions
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
0
Required
0
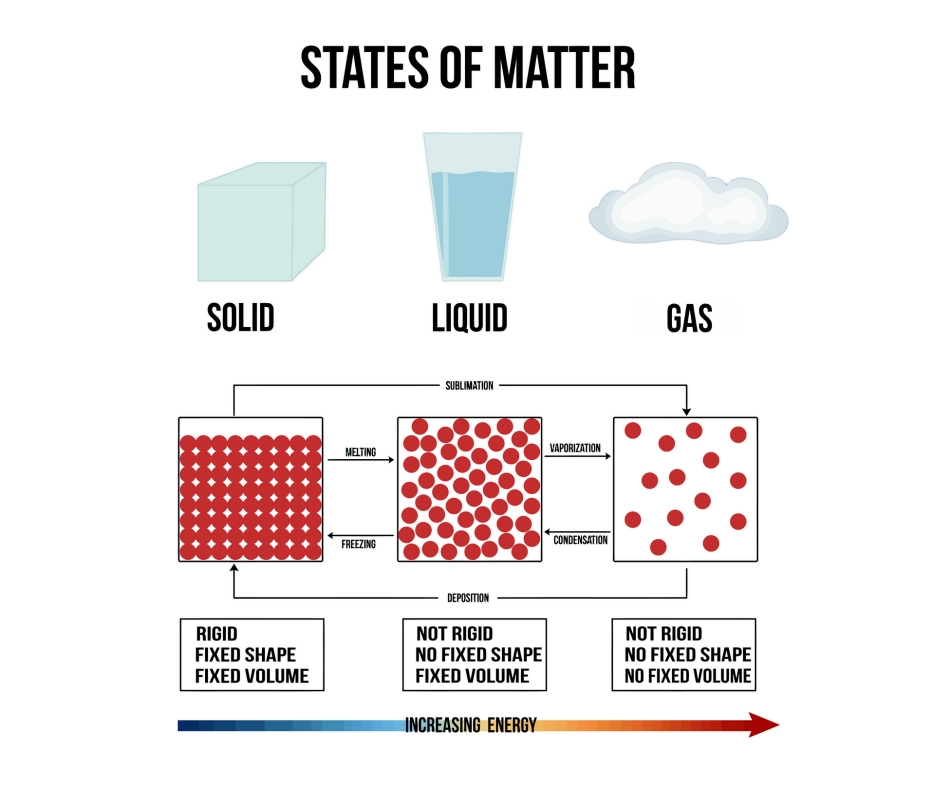
| Draggable item | arrow_right_alt | Corresponding Item |
|---|---|---|
| arrow_right_alt | ||
| arrow_right_alt | ||
| arrow_right_alt |
| Draggable item | arrow_right_alt | Corresponding Item |
|---|---|---|
| arrow_right_alt | ||
| arrow_right_alt | ||
| arrow_right_alt |