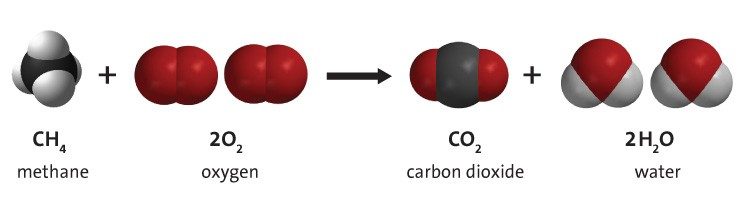
Chemical changes are fascinating events that occur right before our eyes in our everyday world. They involve the transformation of one substance into another, and this process is called a chemical reaction. You might not realize it, but chemical changes are happening around us all the time!
For instance, when you light a piece of wood on fire, a chemical change takes place. That's because the heat causes the molecules in the wood to rearrange and form new substances—like ash, smoke, and gases. Another example is when an iron nail rusts. It reacts with the oxygen in the air, leading to a brand new substance known as rust, which we often notice as a reddish-brown substance on old metal objects.
So, how can we tell if a chemical reaction has taken place? One of the most common indicators is a change in color. Like when a sliced apple turns brown after being exposed to air, that's a clear sign of a chemical reaction! Other hints can include the formation of bubbles (release of gas), the formation of a solid from a liquid (called a precipitate), heat or light being produced, or even a change in smell.
Remember, the key to a chemical reaction or chemical change is the formation of a new substance. Through this, it’s easy to see that molecules are constantly remixing to help create the world we see and experience every day.