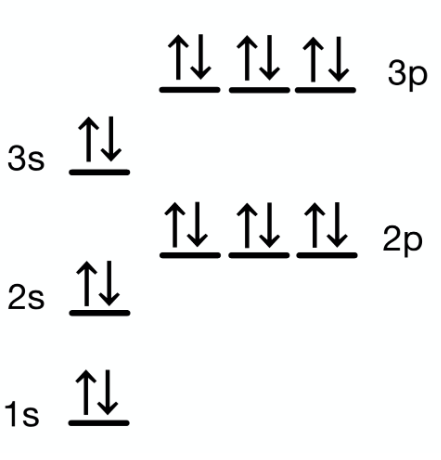
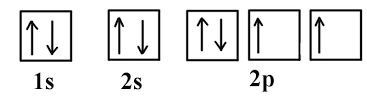
Retake Copy of Honor's Electron Configuration & Orbital Diagrams Quiz (11/22/2024)
star
star
star
star
star
Last updated 12 months ago
18 questions
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1