Integrated Science Fall Final Exam
star
star
star
star
star
Last updated 6 months ago
48 questions
Nature of Science Unit
Required
1
Required
1
Required
1
Required
1
Required
1
2
3
Required
1
Required
2
Required
1
Required
1
Hydrsphere Unit
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
1
Required
1
Required
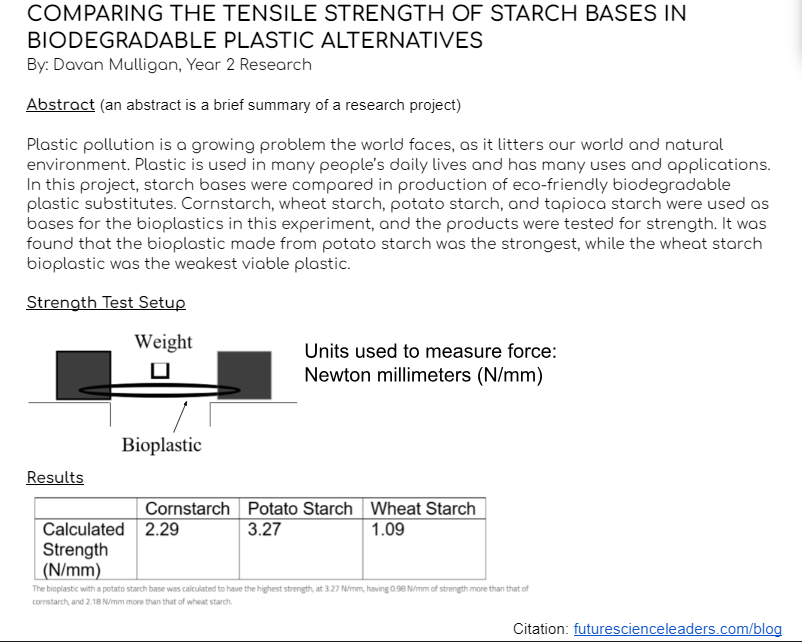
1
Required
1
Chemistry Unit
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
3
Required
1
Required
1
Required
1
Required
2
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1
Required
1