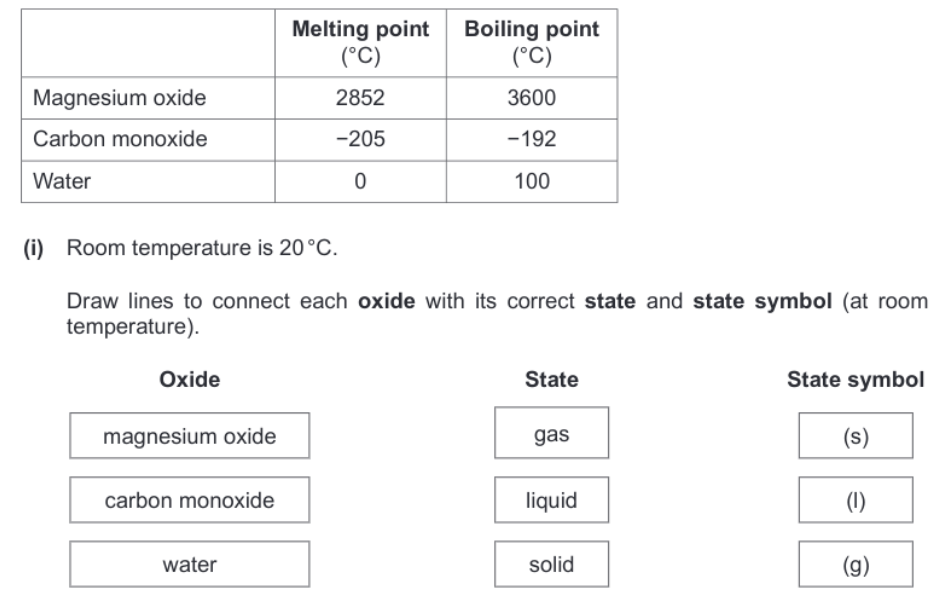
States of matter
star
star
star
star
star
Last updated 10 months ago
15 questions
1
11
13
1
2
1
1
1
3
4
8
1
3
3
3
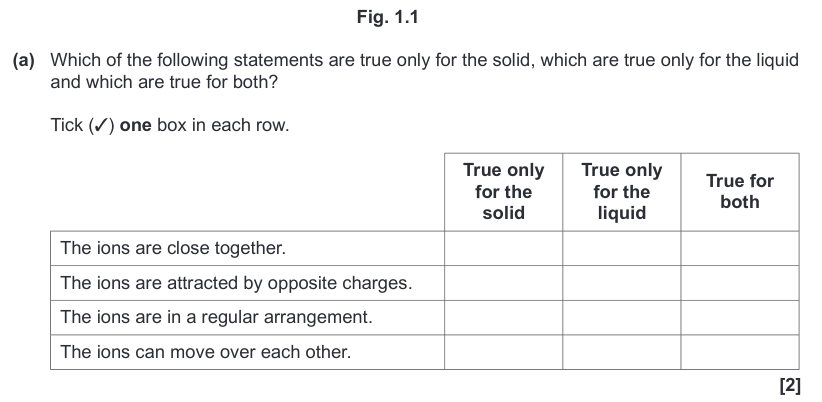




| Draggable item | arrow_right_alt | Corresponding Item |
|---|---|---|
add potassium chloride | arrow_right_alt | mass balance |
measure mass | arrow_right_alt | thermometer |
| arrow_right_alt | ||
| arrow_right_alt |