C8 Gas Tests and Separating Mixtures FT (05/02/2026)
star
star
star
star
star
Last updated 4 months ago
20 questions
4.8.1.1 Pure substances
Required
1
4.8.1.1
Required
1
4.8.1.1
Required
1
4.8.1.1
Required
1
4.8.1.1
2
4.8.1.1
Required
1
4.8.1.1
Required
1
4.8.1.1
1
4.8.1.1
5.5.A
Required
1
4.8.1.1
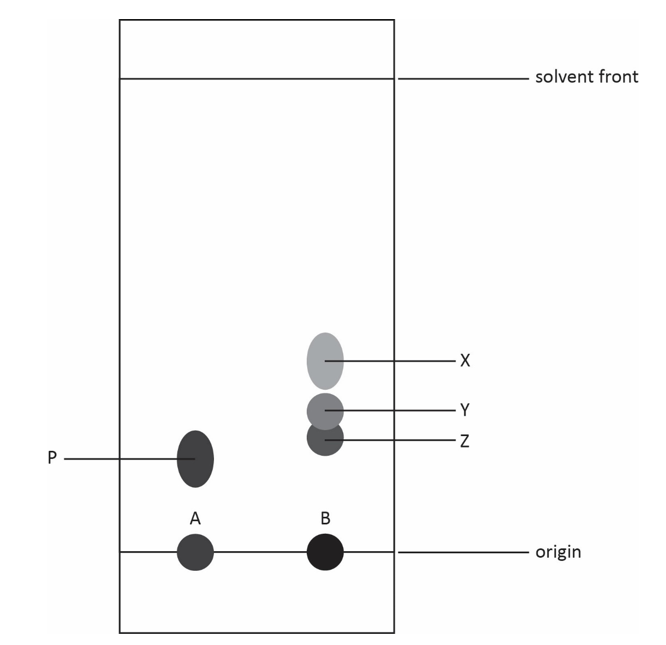
4.8.1.3 Chromatography
1
4.8.1.3
1
4.8.1.3
1
4.8.1.3
1
4.8.1.3
1
4.8.1.3
2
4.8.1.3
8
4.8.1.3
4.8.2 Identification of common gases
1
4.8.2.1
4.8.2.2